WE BREAK GENES TO SOLVE RARE DISEASES.
Our lab sits at the intersection of cell biology, imaging, genome engineering, and rare diseases. We use CRISPR in Xenopus and human cells to engineer disease variants, moving from sequence to mechanism and turning genetic data into biological understanding. Our work uncovers new gene-disease associations, reveals unexpected signaling pathways, and builds the mechanistic evidence that changes how rare diseases are understood and ultimately treated.
MEET THE TEAM
-

Saurabh Kulkarni
Assistant Professor
-

Dana Urbatsch
Lab manager
-

Angelo Arrigo
Graduate Student
-

Savanna Hinson
Graduate Student
-

Venkatraman Rao
Senior Postdoctoral Associate
-

Vani Narayan
Postdoctoral Associate
-

Victoria Hua
Undergraduate Researcher
-

Anburaj Jeyraj
Research Associate
RESEARCH
WHAT IS FUNCTIONAL GENOMICS?
Functional genomics is the science of uncovering how genes actually work in living systems, not just which genes are present, but how they control cellular behaviors, function, and development. By integrating genome engineering, multi-omics, imaging, and computational analysis, we uncover how gene networks build a functioning system and how their disruption leads to diseases. Functional genomics lies at the very center of precision medicine as the bridge between genetic information and truly personalized diagnosis, prognosis, and therapy.
Functional genomics is at the leading edge of biomedical science because it combines big data with deep mechanistic understanding. As a trainee, you will:
Learn state-of-the-art methods like CRISPR screening, high-resolution imaging, omics, and precision disease modeling in Xenopus and human cells.
Work across cell biology, human genetics, and computational biology.
Work on high-impact problems that bridge bench science with real-world medicine.
Be part of a collaborative and interdisciplinary team working to redefine how we study, diagnose, and ultimately treat complex human disease.
PROJECTS
CENTRIOLES, CILIA, AND CYTOSKELETON BIOLOGY.
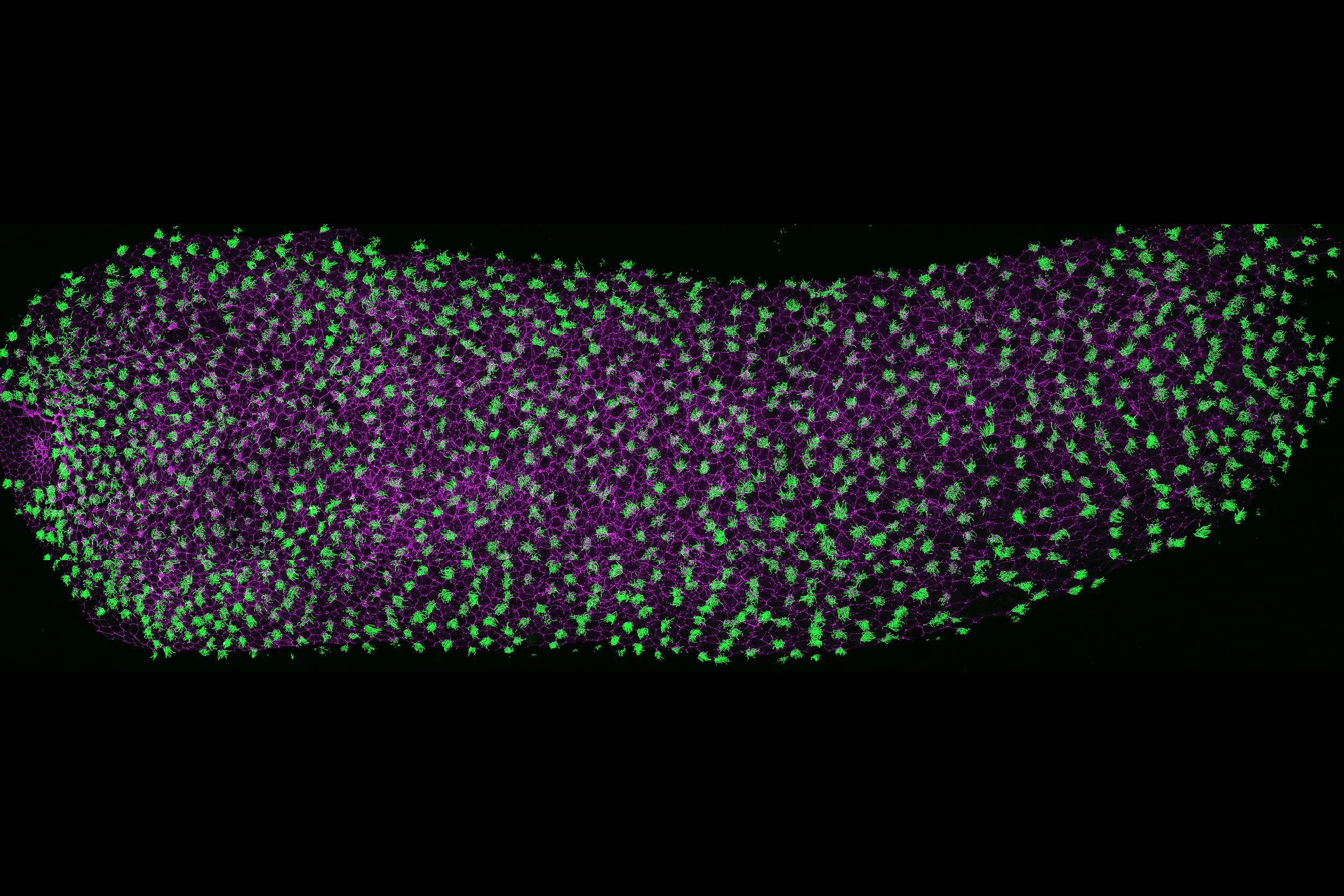
Microtubules play a central role in nearly every cellular process, from cell division and structural organization to intracellular transport and signaling. Our lab focuses on two specialized, microtubule-based structures: centrioles and cilia.
Centrioles are essential for centrosome assembly, organizing the cytoskeleton and mitotic spindles, ensuring accurate cell division and polarity. Cilia, on the other hand, are dynamic, antenna-like projections that are vital for both cellular signaling and motility across tissues. Disruption of either centrioles or cilia can have broad consequences, leading to a range of syndromic disorders, including congenital heart defects, respiratory diseases, metabolic and sensory diseases, and neurodevelopmental disorders such as autism.
We study how centrioles and cilia are assembled, maintained, and regenerated using Xenopus multiciliated cells as our primary model. We combine CRISPR genome engineering, quantitative live imaging, electron microscopy, and mathematical modeling to uncover the molecular rules governing organelle biogenesis.
This fundamental science is the mechanistic foundation for everything else we do.
DNA TO DECISION - IN RARE DISEASES
Genetic sequencing can identify variants. It cannot always tell you what they mean. For families facing a rare disease diagnosis, a "variant of uncertain significance" is not an answer; it is a question mark that can follow them for years.
We take on that question. Using a multi-model functional platform, including Xenopus organism-based screening, human cell validation, and AI-driven structural analysis and machine learning, we systematically test patient variants to determine whether they are disease-causing or benign. Computational predictions help prioritize which variants to test; experimental results improve our models. Our work covers both motile and sensory ciliopathies, addressing a wide range of diseases caused by cilia dysfunction.
This approach has already led to new disease gene discoveries, identified new mechanisms, reclassified variants, and revealed variant-specific clinical outcomes.
If you want to work at the intersection of cell biology, human genetics, and precision medicine, where your experiments directly impact patient diagnoses and clinical outcomes, this is the place.
RESOURCES
MICROSCOPY
NIKON AX-R confocal: This new state-of-the-art microscope is equipped with 8K Galvo and 2K resonant scanners, 4 solid-state laser lines (405, 488, 561, and 640), and 4 PMT detectors with 2 GaAsP detectors. The software capabilities include advanced 2D tracking of the cells or organelles, 3D measurement analysis, deconvolution, and tile scanning.
The LEICA SP8 confocal microscope: The microscope is built on a DMi8 inverted research microscope and comes equipped with a white light laser scan head that is tunable within the range of 470-670nm with up to eight laser lines, a 405nm laser, and a filter-free spectral detector for up to five individual channels.
NIKON SMZ1270 Stereomicroscope: The lab has two sets of stereomicroscopes e, each equipped with a camera and a computer for high-speed imaging.
TOOLS
High-Throughput CRISPR-Cas9 and morpholino-based screening
Light Microscopy: Confocal live and fixed imaging, Super-resolution imaging
Electron Microscopy: Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), and Electron Tomography (ET)
“Omics”: Single-cell/nuclei sequencing and proteomics.
Computational biology: Machine learning and Alphafold structural modeling.
Mechanobiology: Biomechanical manipulations of cells and skin organoids.

Get in Touch
ADDRESS
Department of Cell Biology
Department of Biology
University of Virginia
Charlottesville, VA 22908
PHONE
+1 (434) 297-6833
Affiliations & Programs
Developmental Genomics Center
Bridging developmental biology with genomic and translational science at UVA.
Center for Membrane & Cell Physiology
High-resolution imaging, structural, and biophysical approaches to disease.
Child Health Research Center
Supporting innovative research for childhood diseases
Graduate Programs
BIMS, MSTP, Biology, and T32 training grants